Difference between Galvanizing and Electroplating
Galvanizing and electroplating are both used to protect certain metals against corrosion but they do so in different ways.
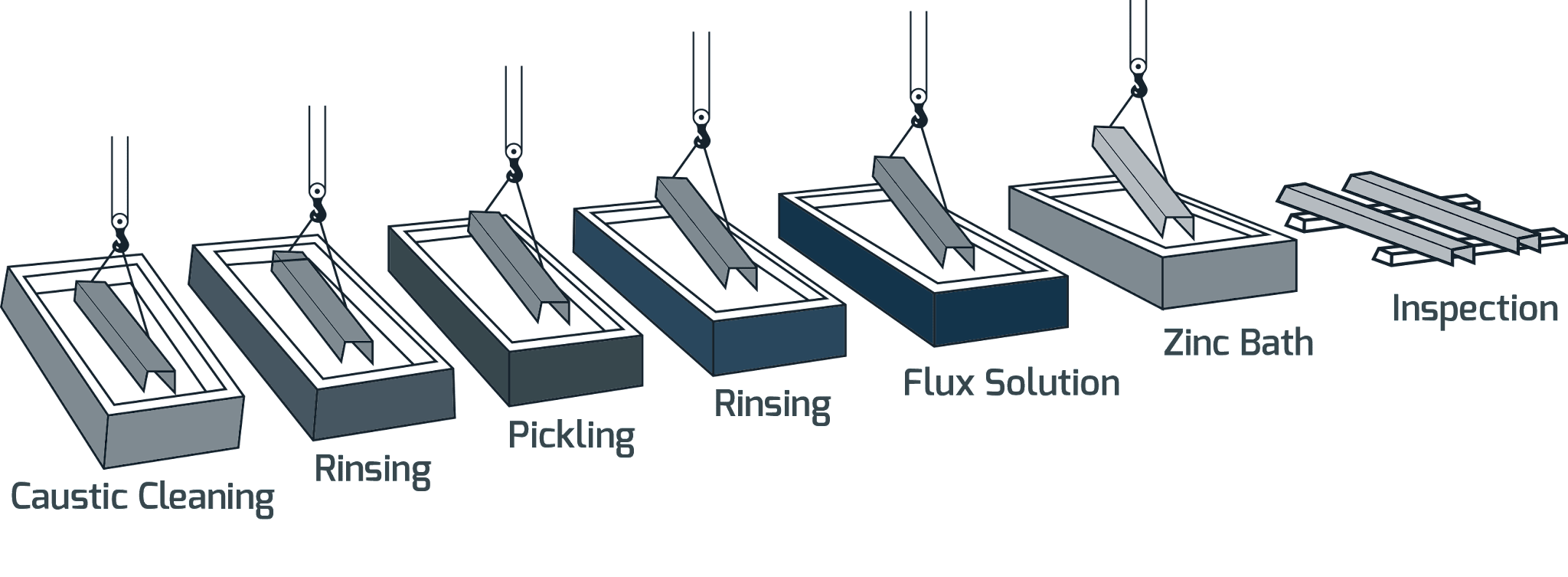
Galvanizing is a metal finishing process that creates a surface layer of zinc on iron or steel.
Electroplating is the process of depositing metal onto a surface through electrolysis or chemical reduction of an alloy. This usually takes place in an electrolytic cell, but can also be done in other ways such as by physical deposition (where the metal is melted and poured into moulds) or even by fusing the metal to its substrate. The electroplating process involves two electrodes: one positive and one negative, which are usually made from different metals.
Galvanization is often used in the making of parts for cars, airplanes, and other equipment that are made from steel.
Galvanizing uses zinc oxide whereas electroplating uses copper sulfate (CuSO4) or silver chloride (AgCl).
Electroplating is a process that creates a thin layer of metal on an object by applying electricity to it. It can be done in different ways such as plating, dipping, and spraying processes. The result is an attractive metallic appearance that will last longer than just using paint or another finished product over bare metal.
Galvanization does not use plating baths, instead, it uses an electrical current that provides oxidation at one electrode while blocking corrosion at the other.
Galvanization increases the strength of the metal, however, it has been criticized for being toxic as well as damaging other metals that come into contact with it.
Electroplating increases the hardness of the metal without affecting its surface finish or machinability.
Galvanization is less time-consuming.
Galvanized products can be reused many times without any loss in performance.
Galvanized produces a smoother surface than electroplating.
Electroplating provides longer-lasting protection
Galvanizing